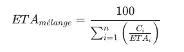Training: Safety Data Sheet (SDS) Authoring and WHMIS 2015 Classification
Master the Art of Compliance: From Chemical Characterization to SDS Publication
The transition to WHMIS 2015 (2022 Revision) is now complete. As of December 2025, all Canadian suppliers and manufacturers must comply with the new requirements. This major update integrates the GHS 7th revised edition and adopts key elements from the 8th edition (GHS 8).
This intensive training is designed to transform your teams into experts in writing safety data sheets (SDS) and in classifying according to the Hazardous Products Regulations (HPR).
Why Choose This Combined Training?
While most training sessions are limited to hazard recognition, Kalium Solutions offers a practical immersion into the regulatory lifecycle of a chemical product.
- GHS 7 & 8 Compliance: Integrate new hazard classes, such as Chemicals Under Pressure and Flammable Gases Category 1B.
- Authoring Autonomy: Reduce outsourcing costs by learning to produce your own SDS.
- Technical Precision: Learn to perform toxicity calculations for mixtures (ATE/ETA).
- North American Harmonization: Learn to author hybrid SDS compliant with both WHMIS 2015 (2022 Rev.) and the new U.S. OSHA HCS 2024 standard.
Don’t just understand WHMIS, master it
| Features | Standard WHMIS Training | Kalium Training |
| Target Audience | General Workers / End-Users | Chemists, R&D, and HSE Managers |
| Primary Focus | Reading and Recognition | Classification & SDS Authoring |
| Update Level | Brief Overview | In-depth GHS 7 and GHS 8 analysis |
| Mixture Calculations | Not Addressed | Full Mastery of ATE Formulas |
| Trade Secrets | Not Addressed | CBI (HMIRA) Claims & Cost Analysis |
| Technical Expertise | General Health & Safety Trainer | Regulatory Affairs Chemists |
| Practical Application | None | Authoring Workshop (Real-world cases) |
| Export Readiness | Canada Only | Canada / USA (OSHA) / Europe (CLP) |
Training Program
Part 1: Advanced Classification
- Classification Principles: Criteria for the 32 physical and health hazard classes.
- Mixture Evaluation: Use of regulatory classification thresholds, extrapolation principles (dilution, interpolation, substantially similar mixtures, production batches, increased in concentration of a hazardous ingredient and aerosols) and acute toxicity calculation formulas.
- GHS 7 and 8 Updates: New categories for aerosols, combustible dusts, and chemicals under pressure
Focus: The Science Behind Mixture Classification
Authoring an SDS is more than just copying and pasting data. Our training delves into complex calculation methods for mixtures, essential for Sections 3 and 11:
- Acute Toxicity Estimate (ATE): Master the calculation formula to classify your products without animal testing.
![ATE acute toxicity calculation formula for WHMIS 2015 mixtures]()
- Bridging Principles: Learn when and how to use data from similar products to justify a classification.
- Weight of Evidence: Knowing how to decide when scientific data is divergent or contradictory.
Part 2: SDS Authoring (16 Sections)
- Regulatory Structure: In-depth analysis of each section required by Health Canada.
- Sections 2 & 3: Authoring Hazard Statements (H), Precautionary Statements (P), and Confidential Business Information (CBI/HMIRA).
- Technical Sections (9, 11, 12): How to transform laboratory data into coherent regulatory text.
Part 3: Labeling and Publication
- Labeling Elements: Create compliant supplier and workplace labels.
- Authoring Tools: Demonstration of process optimization via software solutions.
Crucial Note on Compliance: With the transition period ending in December 2025, any SDS not updated to the 2022 revision (GHS 7/8) exposes your company to non-compliance risks during Health Canada inspections.
Training Plan
Module 1: Introduction
Module objectives: be able to:
- Linking the path of UN recommendations to WHMIS 2015 (rev. 2022)
- Understand how CBI requests work
1.4 History
1.4.1 Globally Harmonized System
1.4.2 WHMIS 2015
1.5 Confidential Business Information (CBI)
1.5.1 History
1.5.2 Founctionning within WHMIS
1.5.3 Application Requirements
1.5.4 Associated costs
1.6 WHMIS Application
1.6.1 WHMIS-exempt products
1.6.2 Hierarchy of Hazards
1.6.3 Change in classification methodology
Module 2: WHMIS Classification of Physical HazardsModule objectives: be able to:
2.21 Explosives |
 |
Module 3: WHMIS Classification of Health Hazard
Objective of the module: to be able to:
- Know the tests and criteria for classifying health hazards
- Understanding the Calculation for Acute Toxicity
- Choosing the Right Classification Criteria for Corrosion Based on the Nature of the Product
3.14 Health Hazard Categories
3.15 Classification Steps
3.15.1 Extrapolation
3.16 Acute toxicity
3.16.1 Key considerations of acute toxicity — inhalation
• Gas vs vapors vs dust/mist
3.16.2 Toxicity calculation for a mixture
3.17 Corrosives
3.17.1 Skin corrosion / irritation
3.17.2 Severe eye damage / eye irritation
3.17.3 Classification for aqueous solutions
3.17.4 Classification for organic solutions (Expert)
3.18 Respiratory or skin sensitization
3.19 Germ cell Mutagenicity
3.20 Carcinogenicity
3.20.1 Classification of a mixture
3.20.2 Differences between Canada and the U.S.
3.20.3 Databases : IARC vs ACGIH vs NTP
3.21 Reproductive toxicity
3.22 Specific target organ toxicity — single exposure
3.22.1 Classification of a mixture
3.22.2 Particularity of Category 3
3.23 Specific Target Organ Toxicity — Repeated Exposures
3.23.1 Classification of a mixture
3.24 Aspiration Hazard
3.25 Biohazardous Infectious Materials
3.26 Health Hazards Not Otherwise Classified
Module 4: Safety Data Sheets (SDS) Authoring
Objective of the module: to be able to
- Know SDS requirements
- Write consistent SDSs
4.4 Safety Data Sheet
4.4.1 16-part structure of the SDS
4.4.2 Requirements
• Concentration ranges
4.4.3 Most common non-compliances
4.5 Writing Workshop (Case Study)
4.5.1 Classification of Mixtures
4.5.2 Pitfalls to Avoid and Standardized Phrases (H and P)
4.5.3 Writing Tools
4.6 Conservation of documents
Module 5: WHMIS Labelling
Objective of the module: to be able to
- Know SDS and labelling requirements
- Differentiating between supplier label and workplace label
5.4 Supplier’s Label
5.5 Workplace Label
5.6 Link between the label and the SDS
Module 6: Conclusion
5.7 Europe — CLP
5.7.1. Differences with WHMIS classification
5.8 Training
5.9 Fines and penalties
Who is the SDS Authoring Training For?
| Profile | Key Benefit |
| Chemists & R&D | Ensure compliance right from the formulation stage. |
| HSE Specialists | Audit and validate SDSs received from suppliers. |
| Regulatory Affairs Managers | Guarantee the company meets the latest GHS updates. |
Exporting: Harmonization Between Canada, USA, and Europe
While GHS is global, regional variations persist. This training helps you navigate different jurisdictions to create "multi-country" SDSs:
- Canada (WHMIS 2015 – 2022 revision): Focus on Health Canada requirements, consumer product exemptions, and trade secret protection (HMIRA).
- USA (OSHA HCS 2024): Understand the subtleties of Hazards Not Otherwise Classified (HNOC) and labeling differences.
- Europe (REACH & CLP): Introduction to specific European format requirements and Exposure Scenario annexes (extended SDS).
Logistics and Registration
- Format: On-site, remote via videoconference.
- Included: Comprehensive training materials, writing SDS and classification for mixture exercises, and a certificate of completion
- Expertise: raining led by chemists specializing in safety data sheet writing and regulatory affairs
Ready to take control of your regulatory compliance?
| Request a Personalized Quote |
View our SDS Authoring Software |
Frequently Asked Questions (FAQ)
Why was WHMIS 2015 updated in 2022?
Canada updated the HPR to align with trading partners, specifically the the European Union (CLP) and the U.S.A. (HCS). This update includes GHS 7 and elements of GHS 8, impacting gas/aerosol classification and adding "Chemicals Under Pressure.".
Is basic WHMIS training enough to author an SDS?
Generally, no. Basic training (employee awareness) teaches how to read an SDS. Safety Data Sheets Authoring requires a deep understanding of toxicological data hierarchy, mixture calculation methods, and specific legal requirements. This is the skill gap our training bridges.
What is the deadline for compliance in Canada?
The official transition period ended in December 2025. From now on, all suppliers, manufacturers, and distributors must have updated their classifications and safety data sheets to comply with GHS 7 and certain elements of GHS 8.
Since the deadline has now passed, companies are subject to penalties during Health Canada inspections if their documentation is not compliant. However, until the end of the U.S. transition period (November 2027), Health Canada will be somewhat more lenient. Participating in our training allows you to bring your situation into compliance immediately, understand the nuances between WHMIS and the U.S. HCS 2024 standard, and avoid any disruptions in your supply chain.
Can I author one SDS for both Canada and the USA?
Yes. We teach you how to structure a hybrid SDS that simultaneously satisfies WHMIS 2015 (2022 Rev.) and OSHA HCS 2024, while respecting specific linguistic requirements for Quebec.
Do you provide tools to help with authoring after the training?
Absolutely. We present automation best practices and offer demonstrations of our SDS Authoring Software, designed to generate compliant documents in just a few clicks.
Ready to take control of your regulatory compliance?
| Request a Personalized Quote |
View our SDS Authoring Software |

Design, programming and hosting by VisionW3.com